TAZVERIK® is a prescription medicine used to treat:
- Adults with follicular lymphoma (FL) when the disease has come back or did not respond to treatment, whose tumors have an abnormal EZH2 gene, and who have been treated with at least two prior medicines. Your healthcare provider will perform a test to make sure TAZVERIK is right for you.
- Adults with follicular lymphoma when the disease has come back or did not respond to treatment, who have no other satisfactory treatment options.
The approval of TAZVERIK in these patients is based on a study that measured the percentage of patients whose tumor shrank or disappeared after treatment and how long that response lasted. TAZVERIK is still being studied to confirm these benefits.
It is not known if TAZVERIK is safe and effective in children less than 16 years of age.
In cancer treatment studies, researchers want to know how many patients respond to therapy. This is measured as overall response, or the percentage of patients whose tumors either shrank by at least 50% or completely disappeared. Every individual is different and may respond differently.
In the TAZVERIK clinical study that included patients with histologically confirmed follicular lymphoma when the disease came back or did not respond after receiving at least two prior treatments, with an EZH2 mutation (N=42) and without an EZH2 mutation (N=53):

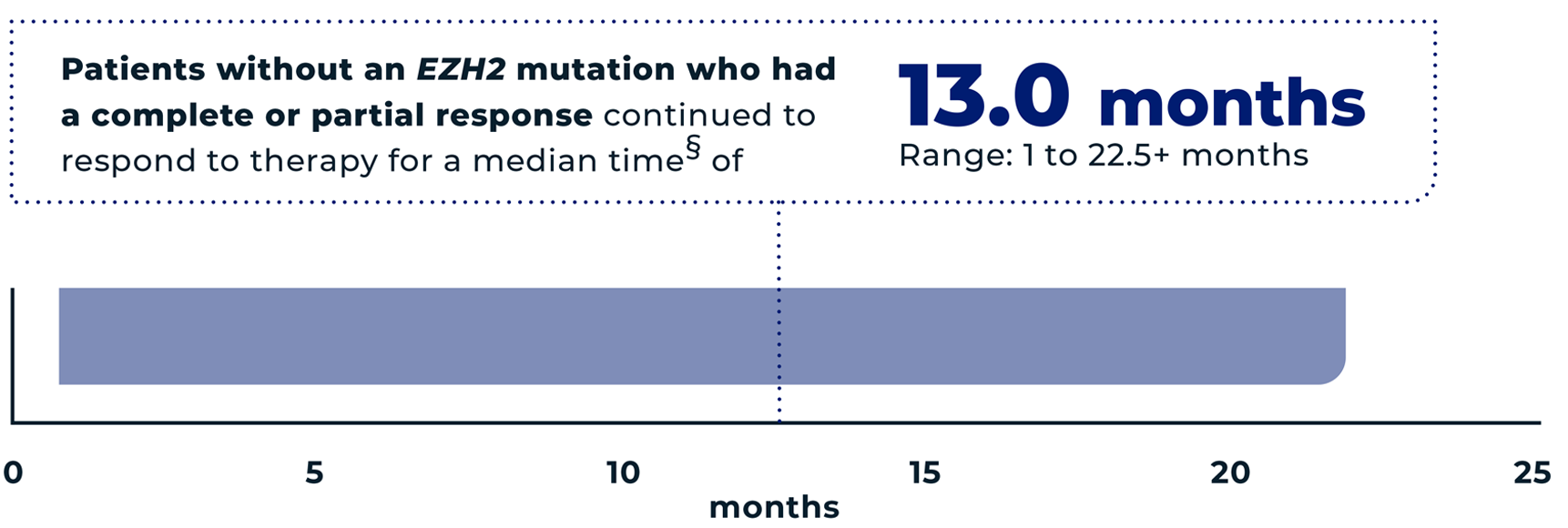
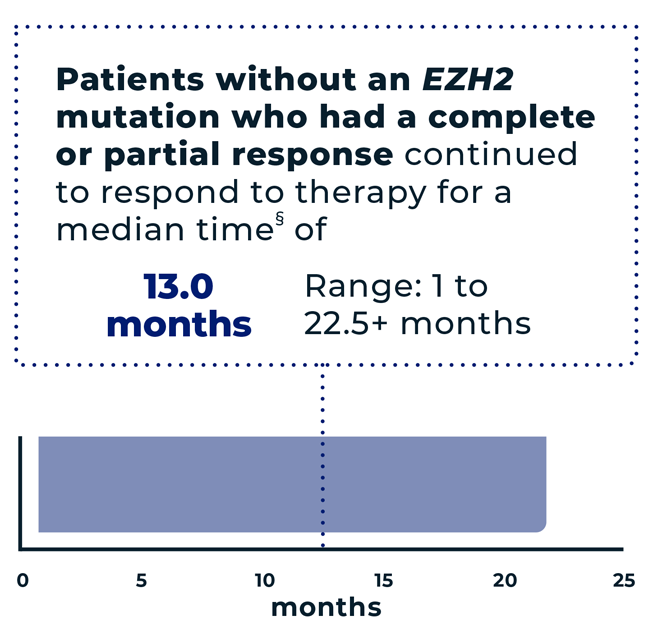
of patients (n=18/53) without an EZH2 mutation had an overall response* to TAZVERIK, including 4% (n=2/53) with a complete response† and 30% (n=16/53) with a partial response‡

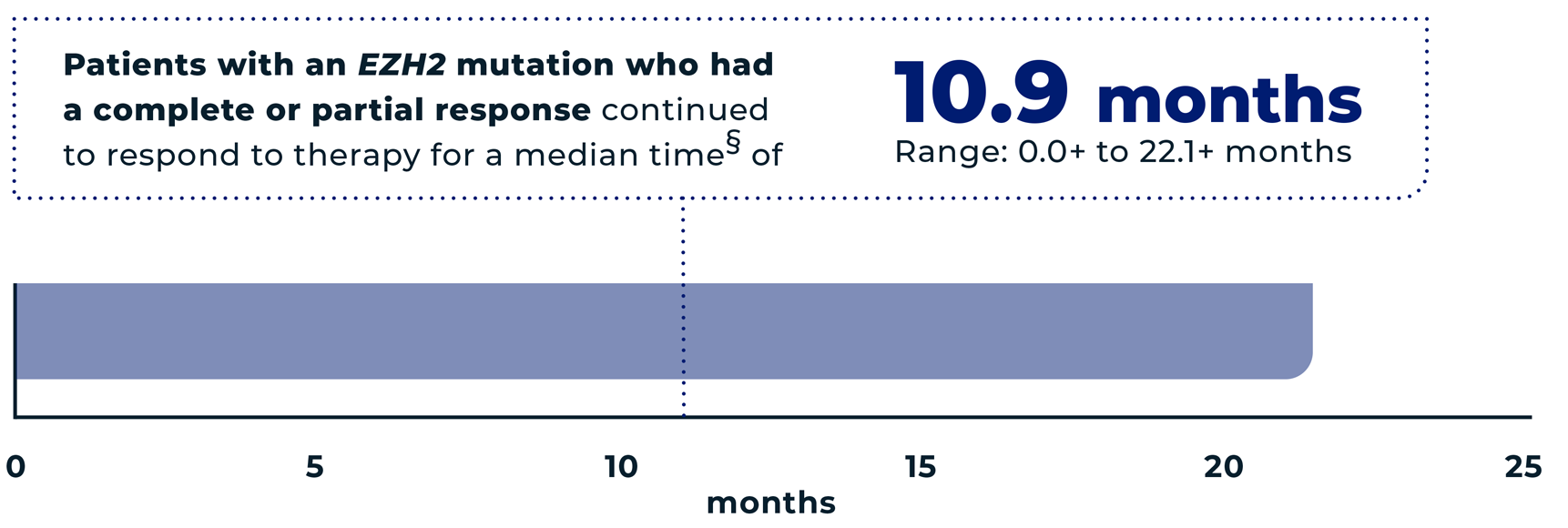
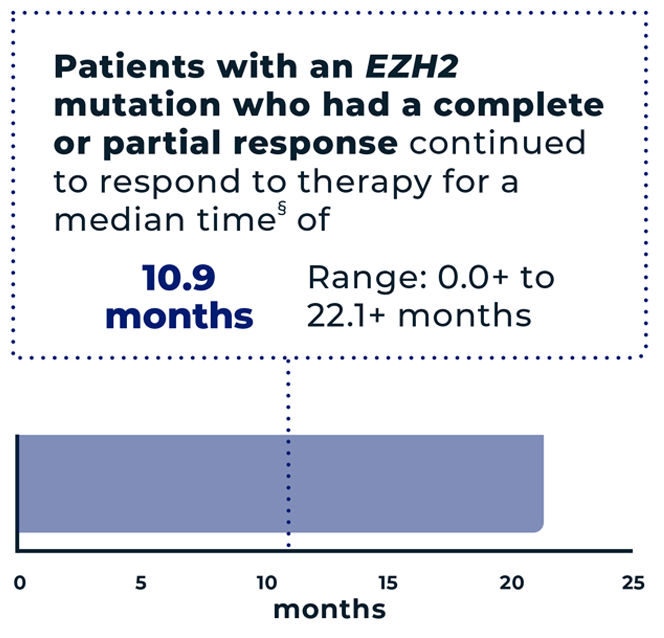
of patients (n=29/42) with an EZH2 mutation had an overall response* to TAZVERIK, including 12% (n=5/42) with a complete response† and 57% (n=24/42) with a partial response‡

The time to achieve a response in the clinical study ranged from:
- around 2 months to around 16 months for patients without an EZH2 mutation, with a median time§ of 3.9 months
- around 2 months to around 11 months for patients with an EZH2 mutation, with a median time§ of 3.7 months

The time to achieve a response in the clinical study ranged from:
- around 2 months to around 16 months for patients without an EZH2 mutation, with a median time§ of 3.9 months
- around 2 months to around 11 months for patients with an EZH2 mutation, with a median time§ of 3.7 months
- *The overall response is the sum of partial responses and complete responses. It means the tumor either reduced in size by at least 50% or completely disappeared.
- †Complete response=the tumor completely disappeared.
- ‡Partial response=the tumor reduced in size by at least 50%.
- §The median is the middle number in a group. In other words, half the patients had a time above the median, and half the patients had a time below the median.
It is important that you are informed about the possible side effects of TAZVERIK. Talk to your doctor about concerns you may have.
What is the most important information I should know about TAZVERIK?
TAZVERIK can cause serious side effects, including:
- Risk of new cancers. An increase in new (second) cancers has happened in people who were treated with TAZVERIK. Talk with your healthcare provider about your risk of developing new cancers. Your healthcare provider will monitor you for new cancers after your treatment with TAZVERIK. Tell your healthcare provider if you are more tired than usual, or have easy bruising, fever, bone pain, or paleness.
What are the possible side effects of TAZVERIK?
The most common side effects of TAZVERIK in people with follicular lymphoma include:
- Tiredness
- Cold-like symptoms (upper respiratory infection)
- Bone and muscle pain
- Nausea
- Stomach (abdominal) pain
These are not all the possible side effects of TAZVERIK. Tell your healthcare provider right away if you have any side effect that bothers you or that does not go away.
You are encouraged to report side effects of prescription drugs to Ipsen Biopharmaceuticals, Inc. at 1-855-463-5127 or to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.

